While the UV systems technology and the appropriate chemistry have been developing continuously, the principle of irradiation curing has widely remained unchanged: High-energy UV irradiation causes chemical curing of UV reactive coatings within seconds.
UV-Spektrum
The short-wave, high-energy UV irradiation in the spectrum between 200nm and 400nm is able to convert a liquid UV reactive substance into a solid film within a split second.
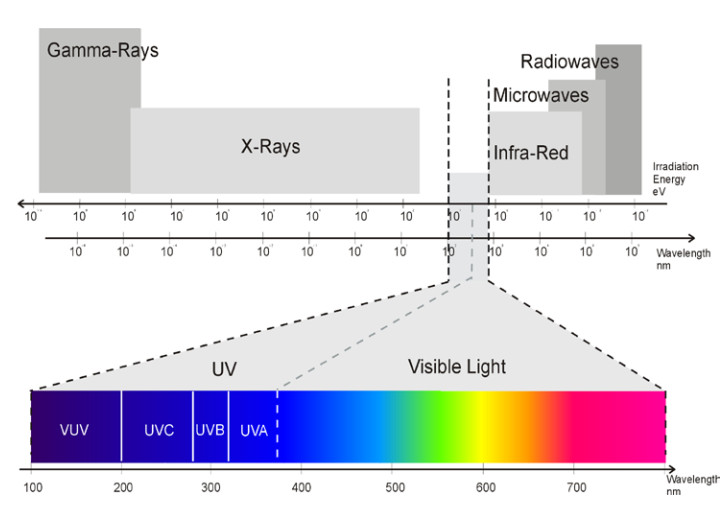
| UV range | Wavelength [nm] |
Photon energy [eV] |
Characteristics | Field of application |
| visible light | 780 - 380 | 1,60 - 3,26 | adhesives | |
| UV-A | 380 - 315 | 3,26 - 3,94 | deep curing | inks or coatings |
| UV-B | 315 - 280 | 3,94 - 4,43 | varnishes | |
| UV-C | 280 - 200 | 4,43 - 6,20 | surface curing | sterilization |
| V-UV | 200 - 100 | 6,20 - 12,4 | absorption by O2 |
no application for UV curing |
Chemical curing
Contrary to a thermal drying process, which works by evaporating the solvent contained in the coating, curing initiates a chemical reaction within the coating compound, which leads to a polymerization reaction.
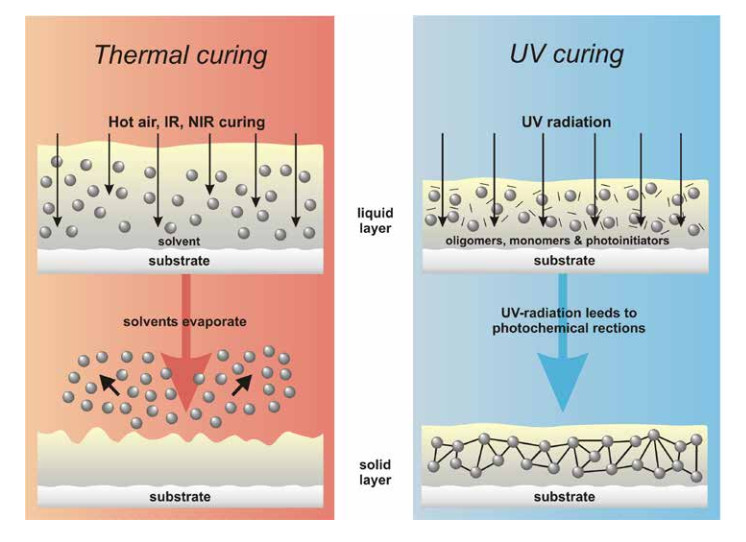
As soon as the reaction is activated by the UV irradiation the fluid layer “cross-links” to an inert flm within a split-second. The majority of UV coatings offer a 100% coating residual, i.e. they cure almost without loss of coat thickness or VOCemissions.
The irradiation-sensitive element of the coating formulation is the photo initiator. Influenced by the UV irradiation, the photo initiator - at a radical polymerization - forms free radicals, which are able to split the double bonds within the oligomeres and monomers. This is the start of a polymerization reaction, which transforms the fluid varnish flm into a three-dimensional structure.







